
It is also important for the function of some globular proteins for example, the hydrophobic residues create a nonaqueous environment that is important for oxygen binding in myoglobin and hemoglobin, and for substrate binding and catalysis in a variety of enzymes ( 3). This is important, not only for the stability of the folded protein, but also for the folding pathway that leads to the mature structure ( 2). Generally, the interior of water-soluble globular proteins consists, largely, of hydrophobic amino acids, principally leucine, isoleucine, valine, phenylalanine, and methionine. This is a crucial determinant of their role in globular proteins, membranous proteins, and coiled-coil structures.

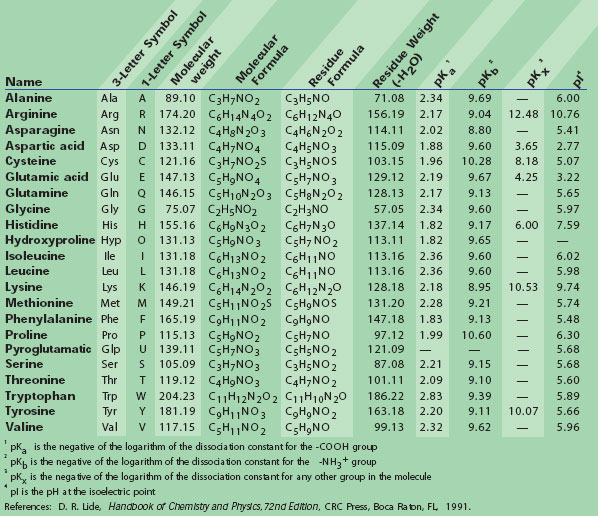
In fact, by some measures of hydrophobicity they are the most hydrophobic ( 1). Leucine, isoleucine, and valine are among the most hydrophobic of amino acids. In this article we discuss the role of the BCAAs in protein structure, their metabolic disposal, the key enzymes of their metabolism, and the regulation of these enzymes. Despite these unusual, or even unique, features it is quite certain that the most important function of these amino acids lies in their roles in proteins. The metabolism of BCAAs presents a number of novel features: the catabolism of these three amino acids is controlled by a common flux-generating step, their catabolic disposal occurs largely in skeletal muscle, their circulating concentrations can influence the brain uptake of precursor amino acids for neurotransmitter synthesis, and they can regulate protein synthesis in a variety of tissues. Leucine, isoleucine, valine, protein structure, muscle metabolism They are thought to play an important role in brain neurotransmitter metabolism. Cytosolic and mitochondrial isoenzymes of BCAT have been identified.
Allosteric inhibition of the kinase by the branched-chain keto acids (BCKA) (particularly by α-ketoisocaproate) serves both as a mechanism for promoting the catabolism of excess quantities of these amino acids as well as for conserving low concentrations of these dietary essential amino acids. BCKD is inhibited by phosphorylation and activated by dephosphorylation. The catabolism of the BCAAs is highly regulated by both allosteric and covalent mechanisms. However, the fact that the flux-generating step for the catabolism of the three BCAAs occurs at one of the common steps indicates that the production of these downstream products are not individually regulated and, hence, may not play important individual roles. Their further metabolism employs distinct pathways to different end-products (glucose and/or ketone bodies). The first steps in their catabolism are common to all three, involving the BCAA aminotransferase (BCAT) and branched-chain α-keto acid dehydrogenase (BCKD). Dietary BCAA largely escape first-pass splanchnic metabolism. These functions are important for almost all proteins therefore, BCAA commonly account for ∼20–25% of most dietary proteins. The occurrence of BCAA in nature is, therefore, attributable to their primary role in protein structure, not to their secondary metabolic roles. Although mutation of one BCAA to another is commonly regarded as conservative, there are well-documented examples of such substitutions that have a significant effect on protein function. In terms of protein secondary structure, valine and isoleucine exhibit a definite preference for the β-structure, whereas leucine has a higher preference for the α-helix. However, the three BCAAs do not behave identically. The three branched-chain amino acids (BCAAs) are the most hydrophobic of the amino acids and play crucial roles in determining the structures of globular proteins as well as the interaction of the transmembrane domains of membranous proteins with phospholipid bilayers.
